Once your assessment is complete, there are a few steps to take before your pharmacy’s accreditation is issued.
View the Quality Care post assessment guide below to familiarise yourself with the next steps of the assessment process
Quality Care 2020 Post Assessment Guide
Viewing your Corrective Actions in the QCPP Customer Portal
Once you have received a copy of your Interim Report (up to 21 calendar days after your assessment week), you will be able to view your pharmacy’s corrective actions in the Customer Portal
As you scroll through your corrective actions, you will see a green, grey, or red circle next to each corrective action.
These indicate the corrective action or remedial action’s status. Refer to the graphic below or see ‘Corrective Action Status’ on the QCPP Customer Portal to check where your tasks are at.

Take me to the QCPP Customer Portal
Corrective Actions
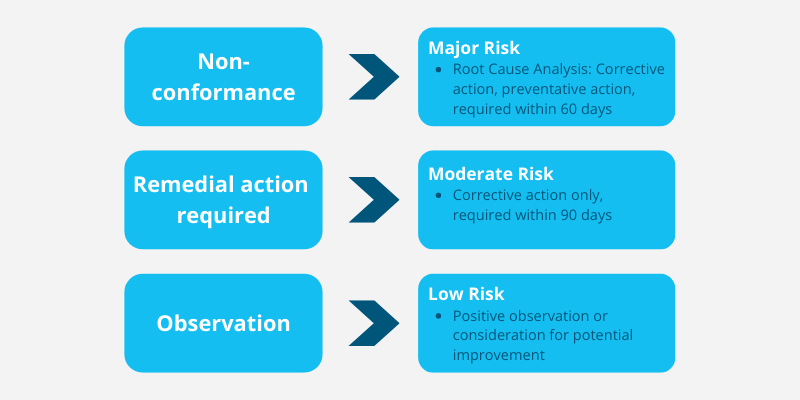
Corrective Actions identify areas where improvements to a pharmacy’s systems, processes or procedures are required to eliminate causes of non-compliance and avoid possible long-term business practice failure.
Corrected actions are divided into three categories (outlined below) based on the level of risk they pose to consumers and the pharmacy business.
Closing out Corrective Actions
It is important to ensure you complete your corrective actions within the 60-to-90-day time frame to avoid lapsing your pharmacy’s accreditation.
Non-Conformances must be completed within 60 days of the interim report being issued, Remedial Actions must be closed out within 90 days.
There is no need for you to take any actions based on Observations feedback, however, it is in the pharmacy’s best interest to consider this feedback
Beginning the Corrective Actions process soon after your interim report is issued gives your pharmacy time to receive support and feedback for evidence submitted during that time.
Redaction
When submitting your corrective actions on the QCPP Customer Portal, it’s important to remove any personal details from the evidence.
Redaction is intended to allow the selective disclosure of information within a document while hiding other parts of the document.
Redacting key elements of a document is an important step in protecting personal data such as names, date of birth, Medicare numbers etc.
Learn more by clicking the button below.
What is a Root Cause Analysis?
A root cause analysis is used to further understand why the non-conformance occurred and identify how it will be prevented in the future.
To successfully complete a root cause analysis, your pharmacy must:
- Provide evidence of action to correct the incident
- Identify why the incident or action occurred; and
- Develop and provide evidence of how the risk will be prevented in the future.
Completing a Root Cause Analysis Fact Sheet
For more support with closing out your corrective actions, complete the Corrective Actions Short Course or visit the Quality Care 2020 Knowledge Hub linked below.
Introducing Corrective Actions Short Course
Quality Care 2020 Knowledge Hub
Common Corrective Actions
CPR and First aid certificates
CPR and First Aid certificates need to be renewed with differing regularity.
CPR Certification must be completed annually to ensure your training meets current standards and your certificate is up to date.
First Aid Certificates are valid for 3 years, but CPR is not included in this.
Cold Chain Certificates and dispensary refrigerator temperature monitoring
You must have dispensary fridge certification listed as a part of your temperature monitoring procedure and ensure that you certify your fridges at least once every 2 years.
S2 S3 training
All pharmacy staff employed who supply S2 & S3 medicines in QCPP accredited pharmacies must complete:
- Initial training in supplying these medicines through a recognised course (within three months of starting employment with the pharmacy)
- Ongoing refresher training of at least three hours per year through a recognised course
For more information, visit S2 & S3 pharmacy assistant training.
Corrective Action Review
Should you wish to dispute receiving a corrective action, you can do so by submitting a Corrective Action Review Request on the QCPP Customer Portal. Your request will be reviewed by the QCPP Interpretations and Rulings Panel within four weeks. If you are not satisfied with the outcome of your Corrective Action Review Request, you may lodge an appeal by emailing help@qcpp.com.
Remedial Action Required Extensions
If you require more time to complete your remedial actions due to extenuating circumstances, you may apply for an extension on the QCPP Customer Portal. Please note this may impact your eligibility for certain CPA payments and contractual arrangements. Please note, extensions are not available for non-conformances as they are higher-risk.
